The Renewal of Dedicated Pharmaceutical Transport Products (PRIO PHARMA)
2026.04.14
Dear Valued Customers,
Thank you for your continued support of the ANA Group.
ANA has previously offered 4 dedicated pharmaceutical transport products. Following the product alignment with Nippon Cargo Airlines (NCA), the pharmaceutical transport product lineup is being renewed. As part of this restructuring, the sales area for PRIO PHARMA - TempAssist (PTA)--a loose cargo product with rapidly increasing demand--is being expanded to all online routes.
Conversely, PRIO MEDICAL will be discontinued. From the effective date, such shipments will be accepted as "General Pharmaceuticals" in compliance with IATA Temperature Control Regulations, with high-quality handling maintained outside the PRIO brand. This reorganization aims to enhance service quality by providing better "clarity" and "ease of selection."
< Note >
1. Commencement Date:
Effective for departures from June 1 (Mon), 2026
(Reservations will be accepted from May 7 (Thu), 2026)
2. Applicable Air Waybill:
ANA Air Waybill (205 prefix)
*Note: For NCA flights (933 prefix), please contact NCA.
*Note: PRIO PHARMA - TempAssist and QuickCare are provided exclusively by ANA.
3. PRIO PHARMA- Product Comparison: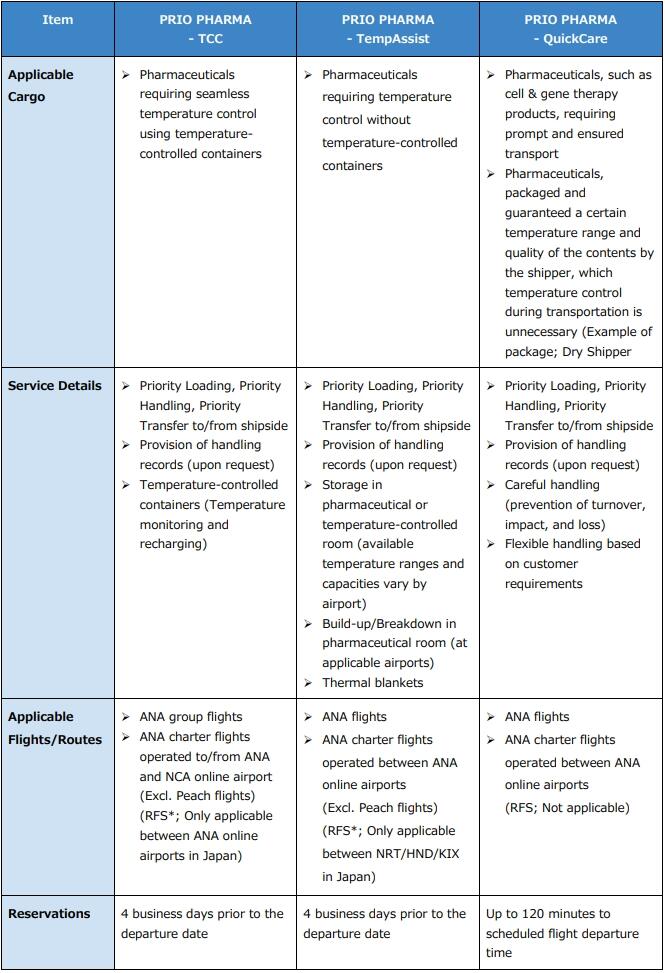
*Road Feeder Service trucks are generally not temperature-controlled. If a temperature-controlled truck within Japan is required, please contact our sales or reservations representative.
4. Points to note when using PRIO PHARMA:
- Airport facility information;
For each airport's facility information, please kindly refer to the following ANA Cargo Website;
-English;
https://www.anacargo.jp/en/int/service/ANA_temp_and_pharma_list_en.pdf
-Chinese; https://www.anacargo.jp/ch/int/service/ANA_temp_and_pharma_list_ch.pdf
-Japanese;
https://www.anacargo.jp/ja/int/service/ANA_temp_and_pharma_list_jp.pdf
For further details, please contact our Sales or Reservations staff in charge.
2. Reservations procedure;
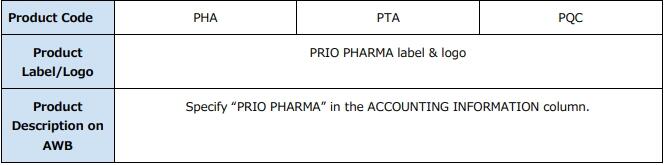
(1) When booking via eSPICA, enter "PHARMA" in the Commodity field and select the appropriate code from the Product field as follows:
PRIO PHARMA - TCC: "PHA" + [Temperature Range (FRO/COL/CRT/ERT)]
PRIO PHARMA - TempAssist: "PTA" + [Temperature Range (FRO/COL/CRT/ERT)]
PRIO PHARMA - QuickCare: "PQC"
(2) In the "Remarks" field, specify the 24-hour available contact (Person in-charge,
Telephone number including the country code, and Email address) for emergencies.
3. Description in the MAWB;
For PRIO PHARMA - TCC and PRIO PHARMA - TempAssist:
(1) Accounting Information column; Indicate "PRIO PHARMA"
(2) Consignee's Name and Address column; Indicate consignee's contact (Person in-charge and Telephone number including the country code).
(3) Handling Information column; Indicate 24-hour available contact (Person in-charge and Telephone number including the country code) for emergencies. In addition, state only one handling temperature range. (Ex.; Keep between +2℃~+8℃ wherever possible.)
For PRIO PHARMA - QuickCare:
(1) Accounting Information column; Indicate "PRIO PHARMA"
(2) Shipper's Name and Address column; Indicate shipper's contact (Person in-charge and Telephone number including the country code and email address).
(3) Consignee's Name and Address column; Indicate consignee's contact (Person in-charge and Telephone number including the country code and email address).
(4) Handling Information column; Indicate 24-hour available contact (Person in-charge and Telephone number including the country code and email address) for emergencies.
4. Points to be noted (Acceptance requirements);
< Packaging >
(1) Wherever possible, storage of the cargo at the airport will be conducted under the specified temperature range. However, depending on each airport's facility conditions or restrictions, the handling under the required temperature may not guaranteed. In order for the cargo to be maintained with the suitable temperature in the package, please ensure that the shipper or forwarder use passive packaging with insulation or coolant, etc., properly.
(2) The shipper or forwarder is responsible to ensure that the product is properly packaged to withstand temperature changes during air transportation.
(3) ANA shall not be liable for any shipment which is damaged and/or loss of commercial value due to inappropriate packaging. Even if the product is properly packed, we do not guarantee that it will be handled under the specified temperatures as described in above "1", and we are not responsible for any damage to the product or loss of product value as a result of such a circumstance.
< Temperature Instructions >
The handling temperature range (※) shall be indicated in the MAWB. Also, please specify "wherever possible".
※+2℃〜+8℃, +15℃〜+25℃, etc.
Ex; Keep between +2℃〜+8℃ wherever possible.
< Temperature Logger >
(1) Only loggers listed in the "Confirmed Device List", approved by ANA, can be loaded on aircraft. For further details, please refer to the following Website;
- English: https://www.anacargo.jp/en/int/regulations/
- Japanese: https://www.anacargo.jp/ja/int/regulations/
(2) The data logger and/or temperature monitor attached to the package of the cargo is easily impacted by the direct sunlight, and it may not be able to measure the temperature of the commodities accurately. For temperature excursions based solely on ambient temperature measurement records may not be accepted as evidence of the commodity's temperature deviation.
For details, please refer to NH in the Operator Variations section of the IATA Temperature Control Regulations. Please note that these requirements also apply to "General Pharmaceuticals".
Should you have any inquiries, please kindly contact our Sales or Reservations staff in charge.
End
【ANA Cargo News】 The Renewal of Dedicated Pharmaceutical Transport Products (PRIO PHARMA).pdf


